Back in July 2016 I wrote about the recommendations made by the UK NSC in March 2015 and why saying no to screening can be a good thing as it means we can avoid harm to people and ensure that money and staff time go to other activities that will bring more benefit.
But as we reiterated at the stakeholder conference last month, the UK NSC members weigh the evidence very carefully and are pleased when evidence of the good that screening can do allows us to say yes.
Such was the case at the November meeting of the UK NSC, when the UK NSC was able to recommend major changes to four existing screening programmes. The UK NSC recommended introducing:
- non-invasive prenatal testing (NIPT) into the fetal anomaly screening programme as part of an evaluative rollout as a more accurate way of identifying pregnant women whose baby has Down’s, Edwards’ or Patau’s syndromes
- faecal immunochemical testing (FIT) to replace the current test used in the bowel cancer screening programme. This is a more accurate test that should help identify and treat more cancers early in their development
- human papillomavirus (HPV) testing as the first stage in the cervical screening programme. This is a better way of identifying women at risk of cervical cancer
- testing for diabetic retinopathy in low risk people every 2 years rather than every year at present. This will free up capacity in the system to better support those people at higher risk without making screening any less effective for those at low risk
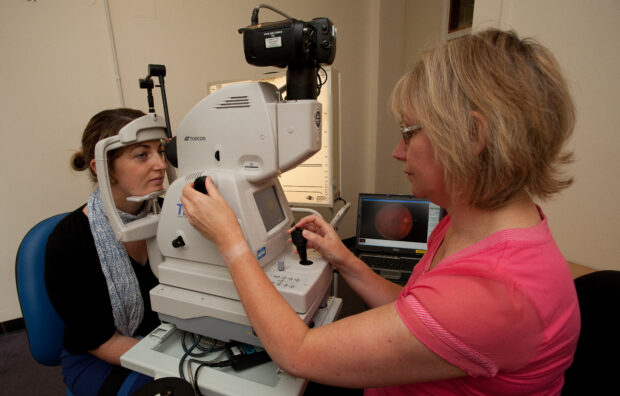
Next steps
So what happens next for these positive recommendations? We are keen for the population benefits of these improvements to be felt as soon as possible and we have already set up expert groups to think through the details so we can co-ordinate the necessary changes as quickly as possible should the recommendations be agreed by the Department of Health and ministers.
These groups have a great deal of work to do in terms of thinking though all the issues around implementation. These include ensuring professionals are trained to deliver new tests, updating public and patient information, thinking through the supporting IT, impact on data collection and reporting, revising clinical pathways, considering impact on doctors and nurses and so on. There’s a lot to do! But we’ve found some great people to help who are not just experts in these fields but passionate champions of screening.
We plan to start putting out regular blogs highlighting the progress made by these groups in the near future.
Other recommendations
At the same meeting, the UK NSC recommended against screening for a number of other conditions. These included congenital adrenal hyperplasia (CAH) in newborns, glaucoma, hearing loss in older adults, mucopolysaccharidosis type I (MPS I), neuroblastoma in children, oral cancer and prostate cancer.
Neuroblastoma (a rare cancer affecting young children) provides an interesting case study in weighing up the pros and cons of screening. A national screening programme was introduced by Japan in 1985 but following evidence from a number of countries that screening may not actually be beneficial, the policy was reviewed in 2003. A special committee found that the screening programme was substantially overdiagnosing neuroblastoma, that is finding tumours that would not have harmed the child (they would have disappeared naturally or become benign). By identifying these tumours through screening, parents were being unnecessarily worried and children were undergoing invasive tests and surgery they didn’t need. So Japan decided to halt the screening programme as there was no evidence it was actually reducing the death rate from neuroblastomas.
I’ll end by touching on the prostate cancer recommendation. This is one of those conditions where there are some understandably strong views that screening should be offered. And evidence does show some benefit from screening. But despite this the harms of treating men who incorrectly test positive still clearly outweigh the potential benefits from screening. The main problem is that prostate specific antigen (PSA) is a very poor test for distinguishing between slow-growing cancers that would never affect a man in his lifetime and fast-growing ones that would. There is lots of research going on into better tests, or improving the accuracy of the PSA test, and if this is successful and all the UK NSC’s criteria can be met then we would be delighted to recommend screening for this condition in the future.
UK NSC blog
The UK NSC blog provides up to date news from the UK National Screening Committee. You can register to receive updates direct to your inbox, so there’s no need to keep checking for new articles. If you have any questions about this blog article, or about the work of the UK NSC, please contact the UK NSC helpdesk.
2 comments
Comment by Jane Raddats posted on
What assistance will Trusts receive to fund the costs associated with the increase in screening tests and workload due to women being screened for sickle & thalassaemia in each pregnancy?
Comment by Cathy Coppinger posted on
Thank for your comment.
No funds are available from the screening programme. This was decided through the Section 7a process and published in NHS public health functions agreement 2016-17 Service specification no.18 NHS Sickle Cell and Thalassaemia Screening Programme:
Definition, identification and invitation of cohort/eligibility P23-24
The target population to be offered screening antenatally is all pregnant women, and the fathers of babies whose mothers are carriers or affected.
In each pregnancy, the family origin questionnaire should be completed and a routine full blood count taken and mean cell haemoglobin (MCH) and other red cell indices should be assessed.
Due to the complexities of the testing algorithm and the logistics of separating samples out new guidance recommends screening for sickle cell and thalassaemia should be repeated in every pregnancy.